UCLA materials scientists and their colleagues have developed a new method to make synthetic biomaterials that mimic the internal structure, stretchiness, strength and durability of tendons and other biological tissues.
The researchers developed a two-pronged process to enhance the strength of existing hydrogels that could be used to create artificial tendons, ligaments, cartilage that are 10 times tougher than the natural tissues. Although the hydrogels contain mostly water with little solid content (about 10% polymer), they are more durable than Kevlar and rubber, which are both 100% polymer. This kind of breakthrough has never been achieved in water-laden polymers until this study, which was recently published in Nature. The new hydrogels could also provide coating for implanted or wearable medical devices to improve their fit, comfort and long-term performance.
“This work shows a very promising pathway toward artificial biomaterials that are on par with, if not stronger than, natural biological tissues,” said study leader Ximin He, an assistant professor of materials science and engineering at the UCLA Samueli School of Engineering.
Hydrogels are a broad class of materials with interior structures made up of crisscrossing polymers or gels. They show promise for use as replacement tissues, either to temporarily close wounds or as a long-term or even permanent solution. In addition, the gels may have applications for soft robots and wearable electronics.
However, current hydrogels are not strong or durable enough to mimic or replace tissues that need to move and flex repeatedly while bearing weight. To address these issues, the UCLA-led team employed a combination of molecular and structural engineering approaches that were not previously utilized together to make hydrogels.
First, the researchers used a method called “freeze-casting” — a solidifying process that results in porous and concentrated polymers, similar to a sponge. Second, they used a “salting-out” treatment to aggregate and crystalize polymer chains into strong fibrils. The resulting new hydrogels have a series of connecting structures across several different scales — from molecular levels up to a few millimeters. The hierarchy of these multiple structures, similar to that of biological counterparts, enables the material to be stronger and more stretchable.
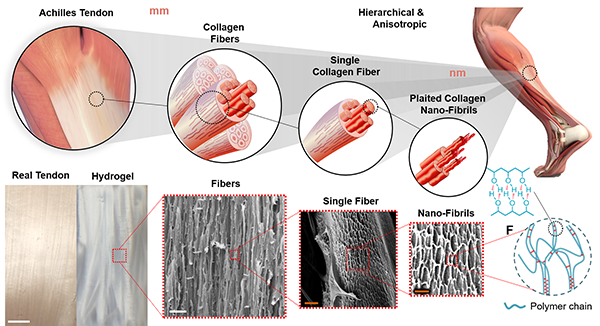
As demonstrated by the team, this versatile method is highly customizable and could replicate various soft tissues in the human body.
The researchers used polyvinyl alcohol, a material already approved by the U.S. Food and Drug Administration, to make their hydrogel prototype. They tested its durability, seeing no signs of deterioration after 30,000 cycles of stretch testing. Under light, the new hydrogel produced a vivid shimmer, similar to real tendons, confirming the micro/nano structures that formed in the gel.
In addition to biomedical applications, the advance may hold potential for surgical machines or bioelectronics that operate innumerable cycles and 3D printing of previously unachievable configuration, thanks to the hydrogel’s flexibility.
In fact, the team demonstrated that such 3D-printed hydrogel architectures could transform into other shapes pending changes in temperature, acidity or humidity. Acting as artificial muscles, they are much more resilient and could exert great force.
The study’s co-lead authors, both from UCLA, are materials science doctoral student Mutian Hua and postdoctoral scholar Shuwang Wu. Other authors include UCLA’s Yanfei Ma, Yusen Zhao, Zilin Chen and Imri Frenkel; Joseph Strzalka and Hua Zhou from Argonne National Laboratory in Illinois; and Xinyuan Zhu from Shanghai Jiao Tong University in China.
The research was supported by the National Science Foundation, Air Force Office of Scientific Research, and the Office of Naval Research. Part of the research was conducted at Argonne National Laboratory, which is operated by the U.S. Department of Energy.

