UCLA Researchers Develop New Method to Customize Artificial Tiny Organs Inside Cells
Advances may lead to specialized biological functions for nanomedicine and gene engineering
Dynamic Nucleic Acid Systems Lab/UCLA
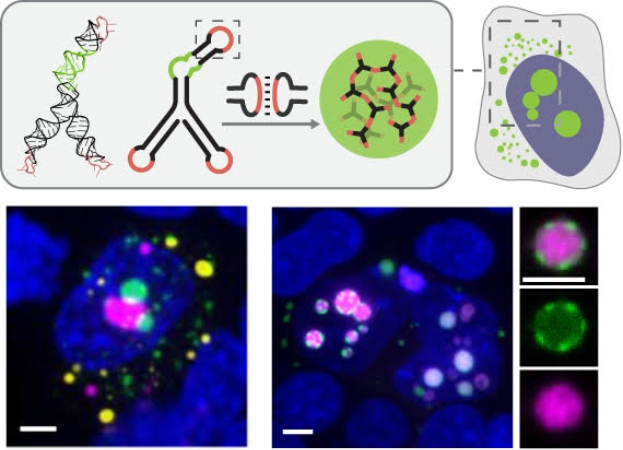
Schematic of UCLA-developed modular RNA motifs forming distinct condensates in cells, shown in different colors at a 5-micrometer scale
UCLA Samueli Newsroom
Just as the human body relies on organs such as the heart or liver for essential functions, cells depend on their own tiny organs, or organelles, to carry out vital tasks, including transporting nutrients, removing waste and regulating genetic activity.
Now, a team of UCLA researchers has developed a new method to build programmable artificial organelles inside living cells using RNA as both the material and the blueprint. The approach enables researchers to design droplet-like cellular compartments that assemble in predictable ways and can be controlled in how and where they form, as well as what molecules they recruit. A study describing the new approach was published today in Nature Nanotechnology.
Some organelles are enclosed by membranes, but others are membrane-less, droplet-like clusters of proteins and RNA known as biomolecular condensates. These structures form as needed and act as temporary workspaces where molecules gather to perform specific functions more efficiently. Artificial condensates have emerged as a promising tool in synthetic biology, offering a way to reorganize the cell’s internal environment and direct chemical reactions and gene activity.
Unlike previous approaches that rely on naturally aggregating proteins, this method encodes assembly instructions directly into the RNA sequence and structure, enabling condensates to be designed with precise interaction rules and tunable properties.
“This research is a step toward architectural engineering of the cell interior,” said study lead Elisa Franco, a professor of mechanical and aerospace engineering and bioengineering at the UCLA Samueli School of Engineering. “By using RNA as a building material, we can create customizable compartments inside cells while using fewer cellular resources than protein-based approaches.”
To drive condensate formation, the researchers designed short strands of RNA that fold into structures they call “nanostars,” each with three or more arms. At the tips of these arms are complementary sequences known as “kissing loops,” which bind to one another and allow the nanostars to assemble into larger networks. Because RNA follows predictable base-pairing rules, the structures can be programmed to form in specific ways.
The team also demonstrated the ability to tune the size, composition and location of the condensates. By modifying the number and length of nanostar arms or the strength of their interactions, the researchers could shift where condensates form within the cell, including between the cytoplasm and the nucleus, where they perform different functions.
“We can control how and where these RNA droplets form and what they attract, effectively creating new, temporary rooms inside the cell furnished with selected molecular tools,” said study first author Shiyi Li, a bioengineering doctoral candidate and a member of Franco’s Dynamic Nucleic Acid Systems Lab.
As the technology develops, these programmable condensates could enable the creation of synthetic organelles inside living cells with specialized biological functions, opening new possibilities in nanomedicine, genetics and cell engineering.
Other authors on the paper include Neil Lin, an associate professor of mechanical and aerospace engineering and bioengineering at UCLA Samueli; Kathrin Plath, a professor of the UCLA Broad Stem Cell Research Center; and associate professor Melody Li and professor Douglas Black, both from UCLA’s Microbiology, Immunology and Molecular Genetics Department. They are joined by Dino Osmanovic, a project scientist in Franco’s group; postdoctoral researchers Anli Tang and Wen Xiao; graduate students Eric Payson, Alexandra Bermudex and Maria Nieto; and undergraduate students Yuna Kim, Kevin Wang, Madison Yang and Diego Dilao.
The research was funded by the National Science Foundation, the Alfred P. Sloan Foundation and the National Institutes of Health. The UCLA Technology Development Group has filed a patent application related to the technology.
